Hydroponic Nutrients for Beginners: What’s Really in the Bottle (and What Plants Actually Use)

Hydroponic nutrients are not “food in a bottle.” They are mostly mineral salts that dissolve into ions, and plants absorb those ions through the roots. Your job is to keep the solution balanced enough that the plant can “drink and eat” without stress.
Key takeaways
- Plants absorb nutrients as inorganic ions, not as “organic chunks.”
- EC tells you “how much total dissolved fertilizer,” not whether the ratio is correct.
- pH influences what stays available, even if your EC looks perfect.
- Two-part nutrients exist because some minerals react and precipitate when stored together in concentrate.
What’s actually in the bottle
Most hydroponic “A/B” nutrients are blends of:
- Macronutrients: nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulfur (S)
- Micronutrients: iron, manganese, zinc, copper, boron, molybdenum (and sometimes others)
Think of it like a well-run kitchen. Macros are the staples, micros are the spices. You do not notice them daily, but missing them ruins the dish.
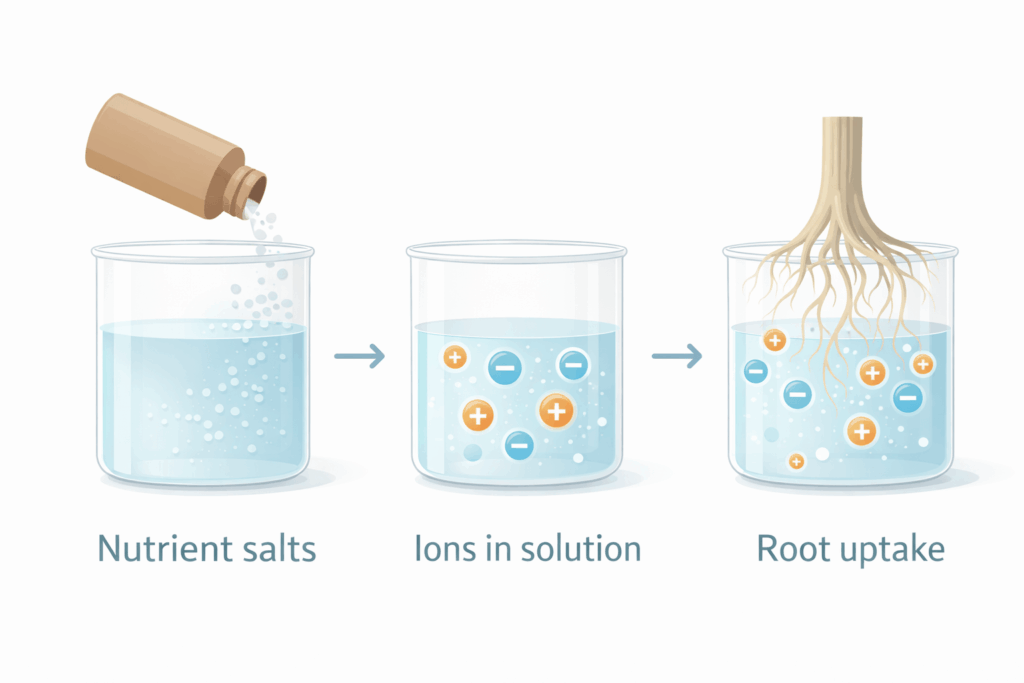
What plants actually absorb
Plants take up nutrients mostly as ions in water (examples):
- N as nitrate (NO3-) or ammonium (NH4+)
- P as phosphate ions
- K as K+
- Ca as Ca2+
This is why hydroponics works: the plant does not need soil to “process” nutrients, it needs ions in solution.

Why nutrients come in Part A and Part B
Concentrated solutions can cause certain combinations to “fall out” as solids (precipitation). The most common incompatibility involves calcium nitrate reacting with phosphates or sulfates in concentrate.
If precipitation happens, those nutrients are no longer available to plants. It is like buying groceries and then locking half of them in a box you cannot open.
Practical rule: Never mix concentrates together. Add each part into a full reservoir (or a bucket of water) separately, with mixing in between.

The 3 numbers that matter (in real life)
- EC (or TDS/PPM): total dissolved salts. A “too high” EC can burn roots; a “too low” EC can starve growth.
- pH: affects availability and uptake. Many hydroponic references put hydroponic pH commonly in the ~5.5–6.5 neighborhood.
- Temperature: affects oxygen and uptake pace (you do not need perfection, you need stability).

A simple mixing workflow (home grower proof)
- Fill reservoir with water.
- Add Part A, circulate 2–3 minutes.
- Add Part B, circulate 2–3 minutes.
- Measure EC and pH.
- Adjust pH if needed (small steps).
- Re-check after 10 minutes of circulation.

If/then troubleshooting (fast)
- If EC is rising week to week and plants look stressed: you are topping off with water that evaporates, salts accumulate. Do a partial change.
- If EC is stable but leaves look deficient: your ratio might be off, or pH is blocking availability. Check pH first.
- If you see white crust or cloudy solution: suspect precipitation or hard-water reactions. Re-mix correctly, consider water quality (next article).
Escalation path
If you have persistent leaf symptoms for 7–10 days despite stable EC and reasonable pH, stop guessing. Take clear photos, note your EC/pH/water source, and consult a local extension resource or a trusted hydroponic advisor.